Versi•SAL®
Saliva Collection Kit
Versi•SAL® kits are for controlled and standardized fluid collection that incorporates a proprietary interchangeable absorbent pad for immediate saliva (and other fluid) collections.
Versi•SAL™ Standard Configuration (Available without tube)
Versi•SAL™ is available in a number of configurations. The “standard” Versi•SAL™ kit includes a plastic compression tube which delivers saliva specimen directly into a standard Eppendorf 2 mL centrifuge tube. A second variation includes a split sample Bifurcating Compression Tube for expressing into 2 sample tubes simultaneously.
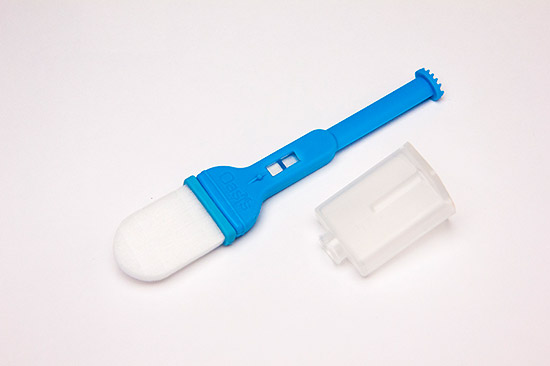
Versi•SAL™-L Transportation Configuration
The Versi•SAL®-L Travel Cover configuration maintains sample integrity for long shipments, thereby relieving the need to remove the sample from the absorbent collection pad at collection point. Further customization can come in thickness of the pad allowing control over maximum sample volume (1.2 mL to 1.4 mL).
Instructions for Use
Versi•SAL® works by placing the device pad under the tongue and collecting saliva (oral fluid) until a novel sample sufficiency indicator (b, above) is triggered [2-3 minutes]. The collector is then pushed down firmly into the compression tube (c) supplied until the Versi•SAL® pad is totally compressed. This action forces the saliva specimen through an outlet (d) into a graduated Eppendorf-style tube (e). The sample obtained may then be tested immediately in point-of-care applications or sent to a laboratory for subsequent testing.

- Take the collection device (a) and place the absorbent pad UNDER the tongue and collect saliva until the sample sufficiency indicator (b) is triggered. At this point the visual line disappears indicating that an adequate volume of saliva has been collected for subsequent analysis, testing or storage for later analysis.
- Left frame above shows the sample sufficiency indicator BEFORE sufficient fluid (saliva) has been collected. Right frame above shows the sample sufficiency indicator AFTER an adequate volume of saliva specimen has been collected.
- Push the Versi•SAL® device (a) down into the compression tube (c) and push until the device can be pushed no further down (the pad will appear totally compressed and saliva will begin to flow into the collection tube.)
- This shows the Versi•SAL® device with fully compressed absorbent pad material. Saliva will be flowing from the sample pad, through the compression tube (d) and into the Eppendorf-style tube (e).
- Remove the Eppendorf-style tube (e) from the outlet of the compression tube (d) and close cap.
- Use the saliva sample collected for immediate (point-of-care testing) applications, send to a qualified laboratory for additional testing or store in a freezer at -20° or -70° Celsius for later analysis.
Instructions for Use
Documentation
Versi•SAL™ Step by Step Instructions
Publications
Saliva: a reliable sample matrix in bioanalytics (M. Gröschl, 2017)
Human Saliva Collection Devices for Proteomics: An Update (Z. Khurshid, 2016)
Salivary Testosterone during the Minipuberty of Infancy (Contreras M. 2017)
Saliva Collection Devices and Diagnostic Platforms (Slowey P. 2015)
Saliva Diagnostics for Oral Diseases (2015)
Acute One-Cigarette Smoking Decreases Ghrelin Hormone in Saliva A Pilot Study (Kaabi Y. 2014)
Salivary Composition and Dental Caries Among Children Controlled Asthmatics (Khalifa M. 2014)
Stress, workload and physiology demand during extravehicular activity: a pilot study (Rai B.2012)
Dehydration decreases saliva antimicrobial proteins important for mucosal immunity (Fortes M. 2012)
In The News

